When we think of wine, we often envision the rich colors, complex aromas, and delightful flavors that make it a staple in celebrations and gatherings. But have you ever stopped to wonder about the composition of this beloved beverage?
Key takeaways
- normally wine is a homogeneous mixture
- natural wines and unfiltered wines tend to be turbid and can therefore be heterogeneous mixtures
- deposits of tartar can form during storage making your wine a heterogeneous mixture
- therefore you can use the state of your wine (homogeneous or heterogeneous) as an indicator on its current condition
Is wine a homogeneous mixture?
Is wine simply a uniform blend of grapes and alcohol, or is wine a homogeneous mixture? Does one substance within it harbor a hidden complexity that challenges our perception of its homogeneity? In this article, we embark on an intriguing exploration into the world of oenology to unravel the enigma: Is wine a homogenous or heterogenous mixture?
The Difference Between Homogeneous and Heterogeneous mixtures?
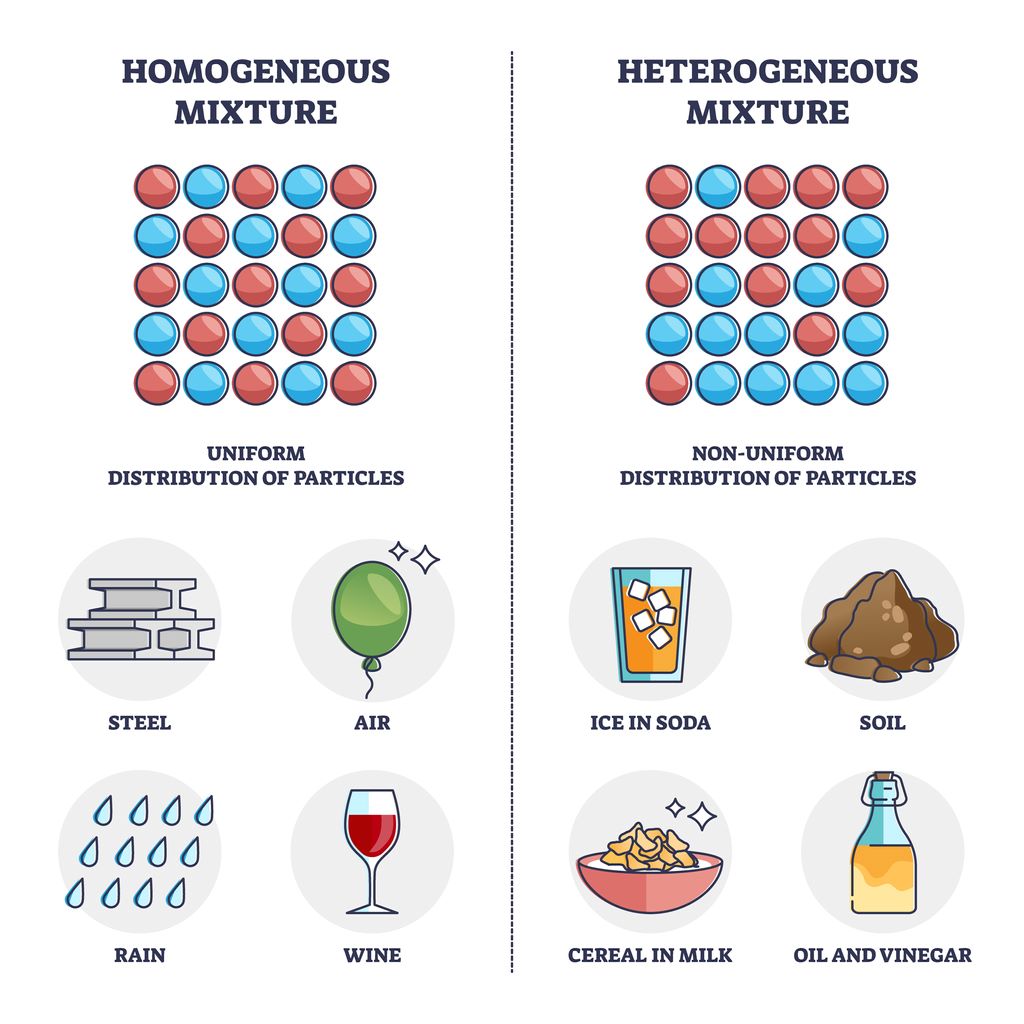
When it comes to mixtures, the terms, homogeneous or heterogeneous mixture are frequently used, but what exactly sets them apart? Is wine a homogeneous mixture?
Homogeneous mixtures
In a homogeneous mixture, the substances are uniformly distributed throughout the solution, resulting in a consistent appearance and composition. This means that no matter where you sample from within the mixture, you will encounter the same combination of substances.

Heterogeneous mixture mixtures
On the other hand, a heterogeneous mixture consist of visibly distinguishable components that do not blend seamlessly together. Whether it’s oil and water or sand and gravel, these mixtures exhibit uneven distribution and variable properties.
Understanding the distinction between two types of homogeneous mixtures and heterogeneous mixtures is crucial in various fields, including chemistry and material science. Homogeneous mixtures play a fundamental role in processes like distillation and filtration due to their uniformity. Meanwhile, knowledge of heterogeneous mixtures is essential for applications such as soil analysis or pharmaceutical formulation where varying compositions can significantly impact outcomes. By comprehending these differences, scientists can make informed decisions about how to manipulate and utilize different types of mixtures for specific purposes.
Why wine is a homogeneous mixture
Wine is often considered a homogenous mixture due to the consistent and uniform appearance, taste, and texture that it presents. A key factor in this homogeneity is the solubility of ethanol in water. Both components are molecularly dispersed throughout the wine, creating a seamless blend that enhances its overall quality and appeal to the palate. This molecular dispersion of alcohol allows for a more even distribution of flavors and aromas, contributing to the harmonious nature of the beverage.

Additionally, during the fermentation process, yeast converts sugar into ethanol and carbon dioxide. This chemical reaction results in a uniform distribution of ethanol throughout the wine, further contributing to its homogeneity. The presence of other organic compounds such as acids and esters also plays a role in creating a cohesive blend with consistent properties. Ultimately, it is this molecular interplay between water, ethanol, and a mixture of other organic compounds that makes wine an intriguing example of a homogenous mixture with its complex yet seamlessly integrated characteristics.
To answer the question is wine a homogeneous mixture, yes it is!
Other Examples of Homogeneous Substances
Homogeneous substances are everywhere in our daily lives, often just unnoticed.
Orange juice and air
Take, for instance, a glass of orange juice, with its consistent texture and color. This is a perfect example of a homogeneous mixture, a substance where the components are thoroughly mixed at the molecular level, ensuring uniformity throughout. Another common example can be found in the air we breathe. Despite being a blend of gases like oxygen, nitrogen, and carbon dioxide, it appears uniform to the naked eye.

Saline and sugar solutions
Moving from liquids to solids, saltwater is another prime example of compound homogeneity. The seamless integration of salt molecules within water forms a solution which remains consistent regardless of where or how it’s sampled. This truly demonstrates the nature of homogeneity at work in our environment. These examples remind us that seemingly simple substances can hold complicated scientific truths about their composition and structure. Other examples are tap water or rain water which contain salts or impurities from the atmosphere.
Natural or unfiltered wine
Wines which are called homogeneous mixtures,can become heterogeneous after longer storage. One key aspect of wine’s heterogeneity is the presence of sediment. It consists of various particles such as tannins, yeast cells, and grape solids. This sediment not only adds complexity to the wine but also serves as a testament to its natural origins.

Another intriguing component of wine’s heterogeneity is tartar, also known as potassium bitartrate. This naturally occurring crystalline substance can be found in unfiltered and minimally processed wines. Its presence not only influences the texture and mouthfeel of the wine but also reflects the winemaking process itself. Embracing these elements of heterogeneity in wine allows us to appreciate the intricate interplay between nature and human intervention in creating this timeless beverage.
Conclusion
Wine can be considered a homogeneous mixture due to its uniform composition at the molecular level. The consistent distribution of water, alcohol, and other components throughout the solution results in a single phase with no visible separation.
After longer storage or considering unfiltered wine it can turn into a heterogeneous mixture. Particles slowly form in the wine and form a sediment after some time.
Understanding wine as a homogeneous mixture helps us appreciate the complexity and balance achieved by winemakers.

